Can Polycythemia Vera Be Caused by Chemical Exposure
The Cardiovascular Organization: Blood
Erythrocytes
Learning Objectives
By the end of this section, you volition be able to:
- Describe the beefcake of erythrocytes
- Discuss the various steps in the lifecycle of an erythrocyte
- Explain the limerick and function of hemoglobin
The erythrocyte, ordinarily known as a red blood jail cell (or RBC), is by far the near common formed element: A single drop of blood contains millions of erythrocytes and just thousands of leukocytes. Specifically, males have nigh 5.4 one thousand thousand erythrocytes per microliter (µL) of blood, and females have approximately 4.8 1000000 per µ50. In fact, erythrocytes are estimated to make up about 25 percent of the total cells in the body. As you tin can imagine, they are quite small cells, with a mean diameter of only almost 7–8 micrometers (µyard) ((Figure)). The chief functions of erythrocytes are to selection upward inhaled oxygen from the lungs and transport it to the torso's tissues, and to selection upwardly some (about 24 percent) carbon dioxide waste at the tissues and ship it to the lungs for exhalation. Erythrocytes remain inside the vascular network. Although leukocytes typically leave the blood vessels to perform their defensive functions, movement of erythrocytes from the blood vessels is aberrant.
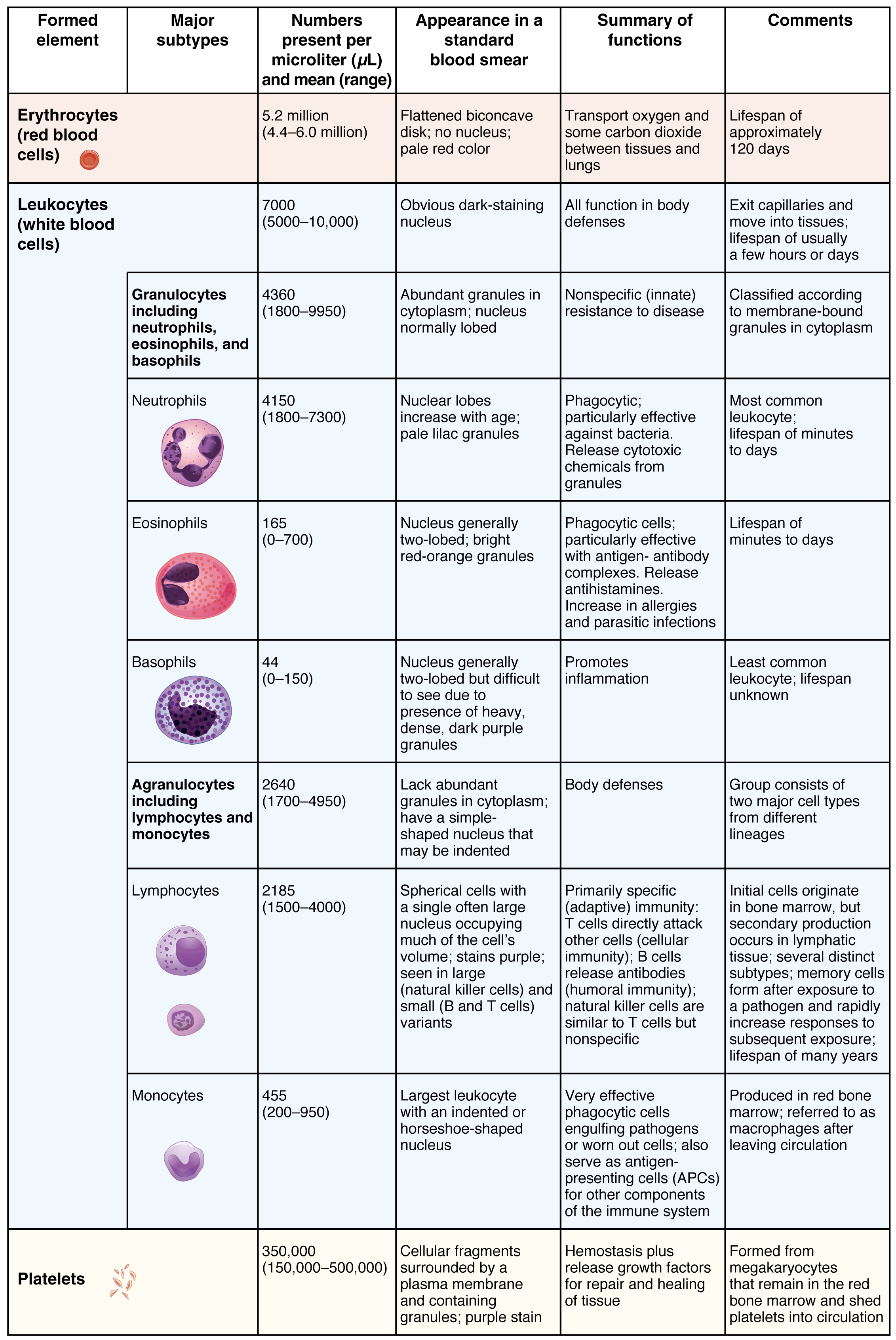
Summary of Formed Elements in Claret
Shape and Structure of Erythrocytes
As an erythrocyte matures in the red bone marrow, it extrudes its nucleus and almost of its other organelles. During the first day or two that information technology is in the apportionment, an young erythrocyte, known as a reticulocyte, will all the same typically contain remnants of organelles. Reticulocytes should contain approximately i–2 percent of the erythrocyte count and provide a rough estimate of the charge per unit of RBC production, with abnormally low or high rates indicating deviations in the production of these cells. These remnants, primarily of networks (reticulum) of ribosomes, are quickly shed, all the same, and mature, circulating erythrocytes have few internal cellular structural components. Lacking mitochondria, for case, they rely on anaerobic respiration. This means that they exercise not utilise any of the oxygen they are transporting, so they tin deliver it all to the tissues. They also lack endoplasmic reticula and do non synthesize proteins. Erythrocytes do, however, comprise some structural proteins that aid the blood cells maintain their unique construction and enable them to change their shape to squeeze through capillaries. This includes the poly peptide spectrin, a cytoskeletal protein chemical element.
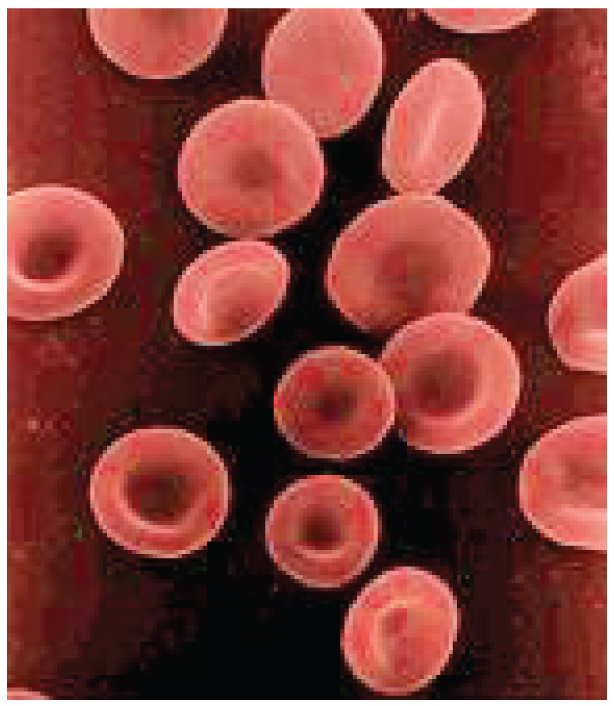
Erythrocytes are biconcave disks; that is, they are plump at their periphery and very thin in the center ((Figure)). Since they lack almost organelles, there is more than interior space for the presence of the hemoglobin molecules that, every bit you volition see shortly, transport gases. The biconcave shape besides provides a greater surface surface area across which gas substitution tin occur, relative to its volume; a sphere of a similar bore would take a lower surface area-to-book ratio. In the capillaries, the oxygen carried by the erythrocytes can diffuse into the plasma and and so through the capillary walls to reach the cells, whereas some of the carbon dioxide produced by the cells every bit a waste diffuses into the capillaries to be picked up by the erythrocytes. Capillary beds are extremely narrow, slowing the passage of the erythrocytes and providing an extended opportunity for gas exchange to occur. However, the space inside capillaries can exist so minute that, despite their own small-scale size, erythrocytes may have to fold in on themselves if they are to make their mode through. Fortunately, their structural proteins like spectrin are flexible, assuasive them to bend over themselves to a surprising degree, so jump back again when they enter a wider vessel. In wider vessels, erythrocytes may stack up much like a ringlet of coins, forming a rouleaux, from the French discussion for "roll."
Shape of Carmine Blood Cells
Erythrocytes are biconcave discs with very shallow centers. This shape optimizes the ratio of surface area to volume, facilitating gas exchange. It likewise enables them to fold up as they motility through narrow blood vessels.
Hemoglobin
Hemoglobin is a large molecule made up of proteins and iron. It consists of four folded bondage of a protein chosen globin, designated alpha 1 and 2, and beta 1 and two ((Figure)a). Each of these globin molecules is bound to a red pigment molecule called heme, which contains an ion of iron (Atomic number 262+) ((Figure)b).
Hemoglobin
(a) A molecule of hemoglobin contains 4 globin proteins, each of which is spring to one molecule of the iron-containing paint heme. (b) A single erythrocyte can comprise 300 million hemoglobin molecules, and thus more than 1 billion oxygen molecules.
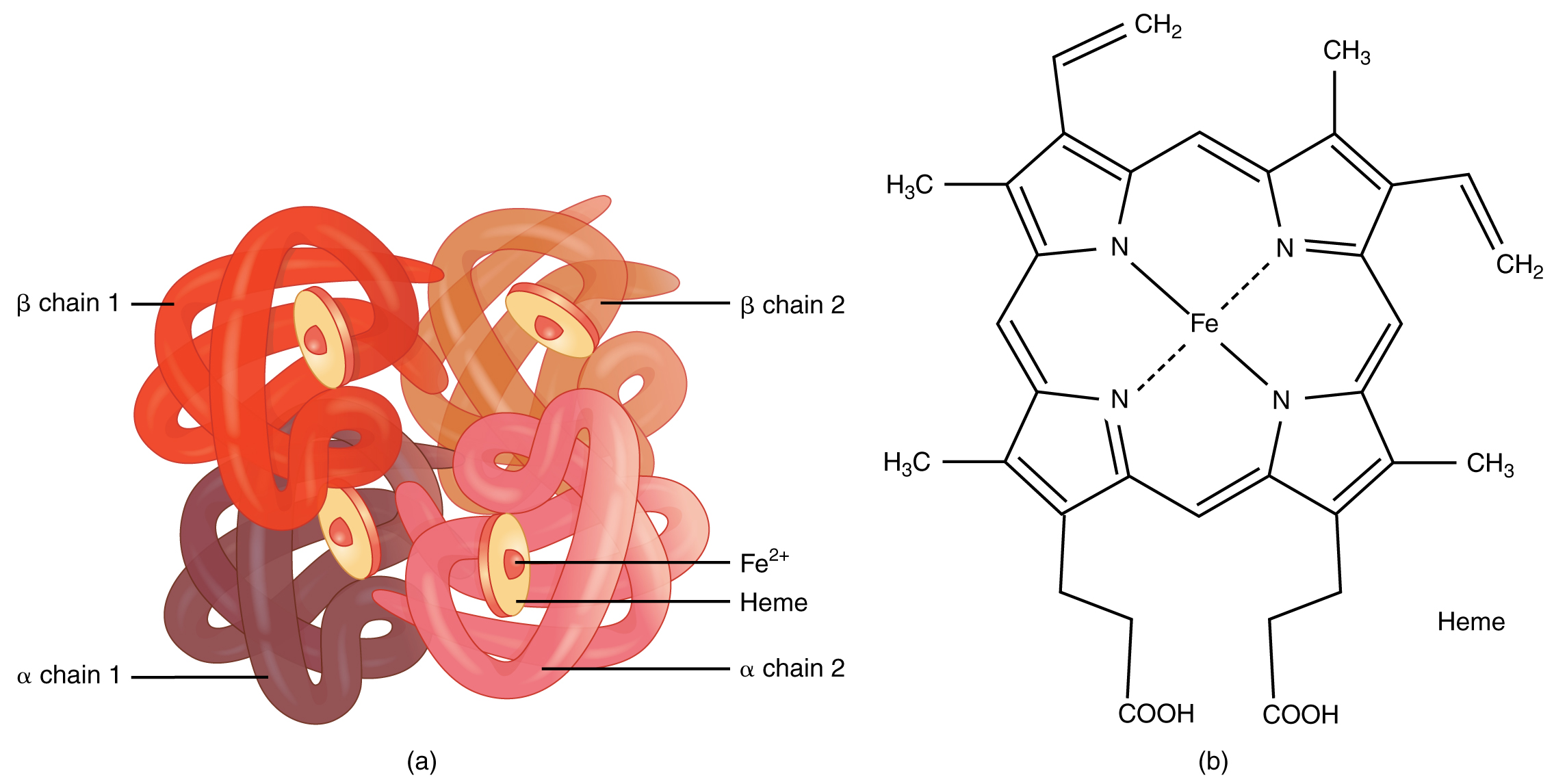
Each iron ion in the heme can bind to one oxygen molecule; therefore, each hemoglobin molecule can transport 4 oxygen molecules. An individual erythrocyte may contain about 300 1000000 hemoglobin molecules, and therefore tin bind to and transport upward to 1.2 billion oxygen molecules (come across (Effigy)b).
In the lungs, hemoglobin picks up oxygen, which binds to the atomic number 26 ions, forming oxyhemoglobin. The bright red, oxygenated hemoglobin travels to the torso tissues, where information technology releases some of the oxygen molecules, condign darker red deoxyhemoglobin, sometimes referred to as reduced hemoglobin. Oxygen release depends on the need for oxygen in the surrounding tissues, so hemoglobin rarely if ever leaves all of its oxygen behind. In the capillaries, carbon dioxide enters the bloodstream. Near 76 percentage dissolves in the plasma, some of it remaining as dissolved COtwo, and the residuum forming bicarbonate ion. Virtually 23–24 percent of information technology binds to the amino acids in hemoglobin, forming a molecule known as carbaminohemoglobin. From the capillaries, the hemoglobin carries carbon dioxide back to the lungs, where it releases it for exchange of oxygen.
Changes in the levels of RBCs tin take pregnant effects on the body's power to finer deliver oxygen to the tissues. Ineffective hematopoiesis results in insufficient numbers of RBCs and results in i of several forms of anemia. An overproduction of RBCs produces a condition called polycythemia. The primary drawback with polycythemia is not a failure to directly deliver enough oxygen to the tissues, but rather the increased viscosity of the blood, which makes it more difficult for the middle to broadcast the blood.
In patients with insufficient hemoglobin, the tissues may non receive sufficient oxygen, resulting in another grade of anemia. In determining oxygenation of tissues, the value of greatest interest in healthcare is the pct saturation; that is, the percentage of hemoglobin sites occupied past oxygen in a patient's blood. Clinically this value is commonly referred to but as "percent sat."
Percent saturation is ordinarily monitored using a device known as a pulse oximeter, which is applied to a thin function of the body, typically the tip of the patient'southward finger. The device works by sending 2 different wavelengths of light (one red, the other infrared) through the finger and measuring the low-cal with a photodetector as information technology exits. Hemoglobin absorbs light differentially depending upon its saturation with oxygen. The machine calibrates the corporeality of low-cal received by the photodetector against the corporeality captivated by the partially oxygenated hemoglobin and presents the data as per centum saturation. Normal pulse oximeter readings range from 95–100 percent. Lower percentages reflect hypoxemia, or depression claret oxygen. The term hypoxia is more generic and merely refers to depression oxygen levels. Oxygen levels are besides directly monitored from free oxygen in the plasma typically following an arterial stick. When this method is applied, the corporeality of oxygen present is expressed in terms of partial pressure of oxygen or simply pO2 and is typically recorded in units of millimeters of mercury, mm Hg.
The kidneys filter about 180 liters (~380 pints) of claret in an average adult each 24-hour interval, or about xx percentage of the total resting volume, and thus serve as platonic sites for receptors that determine oxygen saturation. In response to hypoxemia, less oxygen will get out the vessels supplying the kidney, resulting in hypoxia (low oxygen concentration) in the tissue fluid of the kidney where oxygen concentration is actually monitored. Interstitial fibroblasts within the kidney secrete EPO, thereby increasing erythrocyte production and restoring oxygen levels. In a archetype negative-feedback loop, equally oxygen saturation rises, EPO secretion falls, and vice versa, thereby maintaining homeostasis. Populations home at high elevations, with inherently lower levels of oxygen in the atmosphere, naturally maintain a hematocrit college than people living at ocean level. Consequently, people traveling to loftier elevations may feel symptoms of hypoxemia, such equally fatigue, headache, and shortness of jiff, for a few days subsequently their arrival. In response to the hypoxemia, the kidneys secrete EPO to step up the production of erythrocytes until homeostasis is accomplished in one case again. To avoid the symptoms of hypoxemia, or altitude sickness, mount climbers typically rest for several days to a calendar week or more at a series of camps situated at increasing elevations to allow EPO levels and, consequently, erythrocyte counts to rise. When climbing the tallest peaks, such as Mt. Everest and K2 in the Himalayas, many mountain climbers rely upon bottled oxygen every bit they near the elevation.
Lifecycle of Erythrocytes
Production of erythrocytes in the marrow occurs at the staggering rate of more than 2 meg cells per second. For this production to occur, a number of raw materials must be present in adequate amounts. These include the aforementioned nutrients that are essential to the production and maintenance of any jail cell, such as glucose, lipids, and amino acids. However, erythrocyte product besides requires several trace elements:
- Fe. We take said that each heme grouping in a hemoglobin molecule contains an ion of the trace mineral iron. On boilerplate, less than 20 percent of the iron we consume is absorbed. Heme iron, from animal foods such equally meat, poultry, and fish, is absorbed more than efficiently than not-heme atomic number 26 from plant foods. Upon absorption, atomic number 26 becomes part of the trunk'south total iron puddle. The bone marrow, liver, and spleen can store fe in the protein compounds ferritin and hemosiderin. Ferroportin transports the iron across the abdominal cell plasma membranes and from its storage sites into tissue fluid where information technology enters the blood. When EPO stimulates the production of erythrocytes, iron is released from storage, bound to transferrin, and carried to the ruby marrow where it attaches to erythrocyte precursors.
- Copper. A trace mineral, copper is a component of 2 plasma proteins, hephaestin and ceruloplasmin. Without these, hemoglobin could not be fairly produced. Located in abdominal villi, hephaestin enables iron to be absorbed by intestinal cells. Ceruloplasmin transports copper. Both enable the oxidation of fe from Fe2+ to Fe3+, a class in which it tin be bound to its transport protein, transferrin, for send to body cells. In a state of copper deficiency, the transport of iron for heme synthesis decreases, and iron can accumulate in tissues, where it can eventually pb to organ damage.
- Zinc. The trace mineral zinc functions as a co-enzyme that facilitates the synthesis of the heme portion of hemoglobin.
- B vitamins. The B vitamins folate and vitamin B12 function as co-enzymes that facilitate DNA synthesis. Thus, both are critical for the synthesis of new cells, including erythrocytes.
Erythrocytes live up to 120 days in the apportionment, later which the worn-out cells are removed by a blazon of myeloid phagocytic cell chosen a macrophage, located primarily inside the bone marrow, liver, and spleen. The components of the degraded erythrocytes' hemoglobin are further processed as follows:
- Globin, the protein portion of hemoglobin, is broken downwardly into amino acids, which can be sent back to the bone marrow to be used in the product of new erythrocytes. Hemoglobin that is non phagocytized is broken down in the circulation, releasing alpha and beta chains that are removed from circulation by the kidneys.
- The iron contained in the heme portion of hemoglobin may be stored in the liver or spleen, primarily in the form of ferritin or hemosiderin, or carried through the bloodstream by transferrin to the red bone marrow for recycling into new erythrocytes.
- The non-iron portion of heme is degraded into the waste matter product biliverdin, a green pigment, and then into another waste product, bilirubin, a yellow pigment. Bilirubin binds to albumin and travels in the claret to the liver, which uses it in the industry of bile, a compound released into the intestines to help emulsify dietary fats. In the large intestine, bacteria breaks the bilirubin autonomously from the bile and converts it to urobilinogen and then into stercobilin. It is and then eliminated from the body in the feces. Broad-spectrum antibiotics typically eliminate these bacteria likewise and may alter the colour of feces. The kidneys besides remove whatever circulating bilirubin and other related metabolic byproducts such as urobilins and secrete them into the urine.
The breakup pigments formed from the destruction of hemoglobin can be seen in a multifariousness of situations. At the site of an injury, biliverdin from damaged RBCs produces some of the dramatic colors associated with bruising. With a declining liver, bilirubin cannot be removed effectively from apportionment and causes the torso to assume a yellowish tinge associated with jaundice. Stercobilins within the feces produce the typical brown colour associated with this waste matter. And the yellow of urine is associated with the urobilins.
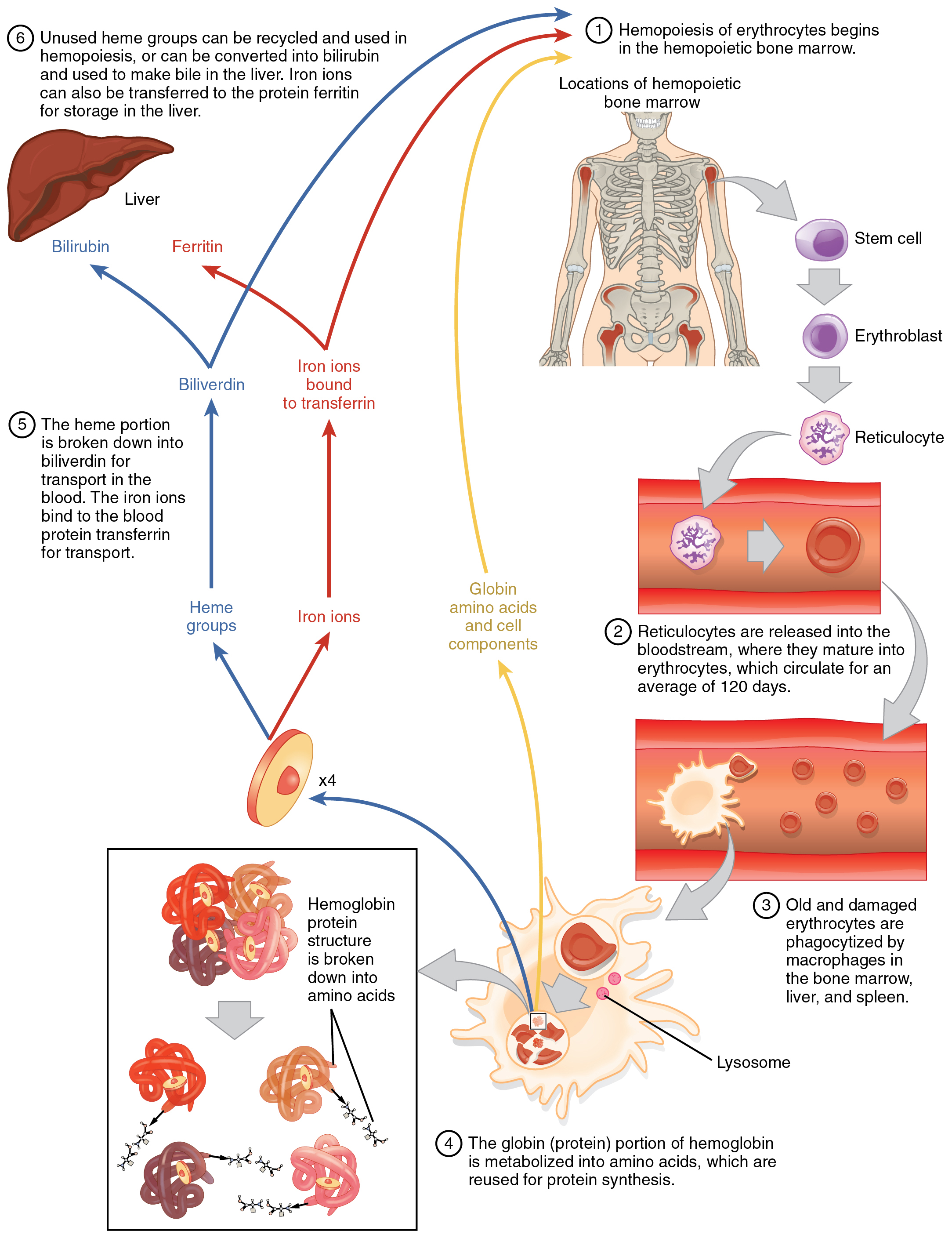
The erythrocyte lifecycle is summarized in (Figure).
Erythrocyte Lifecycle
Erythrocytes are produced in the bone marrow and sent into the circulation. At the end of their lifecycle, they are destroyed by macrophages, and their components are recycled.
Disorders of Erythrocytes
The size, shape, and number of erythrocytes, and the number of hemoglobin molecules tin can have a major impact on a person's health. When the number of RBCs or hemoglobin is deficient, the general condition is chosen anemia. At that place are more 400 types of anemia and more than iii.v meg Americans suffer from this condition. Anemia can exist cleaved downwards into three major groups: those caused past blood loss, those caused by faulty or decreased RBC production, and those caused by excessive destruction of RBCs. Clinicians often employ two groupings in diagnosis: The kinetic approach focuses on evaluating the production, destruction, and removal of RBCs, whereas the morphological approach examines the RBCs themselves, paying item accent to their size. A common exam is the mean corpuscle volume (MCV), which measures size. Normal-sized cells are referred to equally normocytic, smaller-than-normal cells are referred to as microcytic, and larger-than-normal cells are referred to every bit macrocytic. Reticulocyte counts are also important and may reveal inadequate production of RBCs. The effects of the various anemias are widespread, considering reduced numbers of RBCs or hemoglobin will consequence in lower levels of oxygen being delivered to body tissues. Since oxygen is required for tissue functioning, anemia produces fatigue, lethargy, and an increased risk for infection. An oxygen deficit in the brain impairs the ability to think clearly, and may prompt headaches and irritability. Lack of oxygen leaves the patient brusque of breath, fifty-fifty as the heart and lungs piece of work harder in response to the deficit.
Blood loss anemias are fairly straightforward. In addition to bleeding from wounds or other lesions, these forms of anemia may be due to ulcers, hemorrhoids, inflammation of the stomach (gastritis), and some cancers of the gastrointestinal tract. The excessive apply of aspirin or other nonsteroidal anti-inflammatory drugs such every bit ibuprofen can trigger ulceration and gastritis. Excessive catamenia and loss of claret during childbirth are also potential causes.
Anemias caused past faulty or decreased RBC production include sickle cell anemia, iron deficiency anemia, vitamin deficiency anemia, and diseases of the os marrow and stem cells.
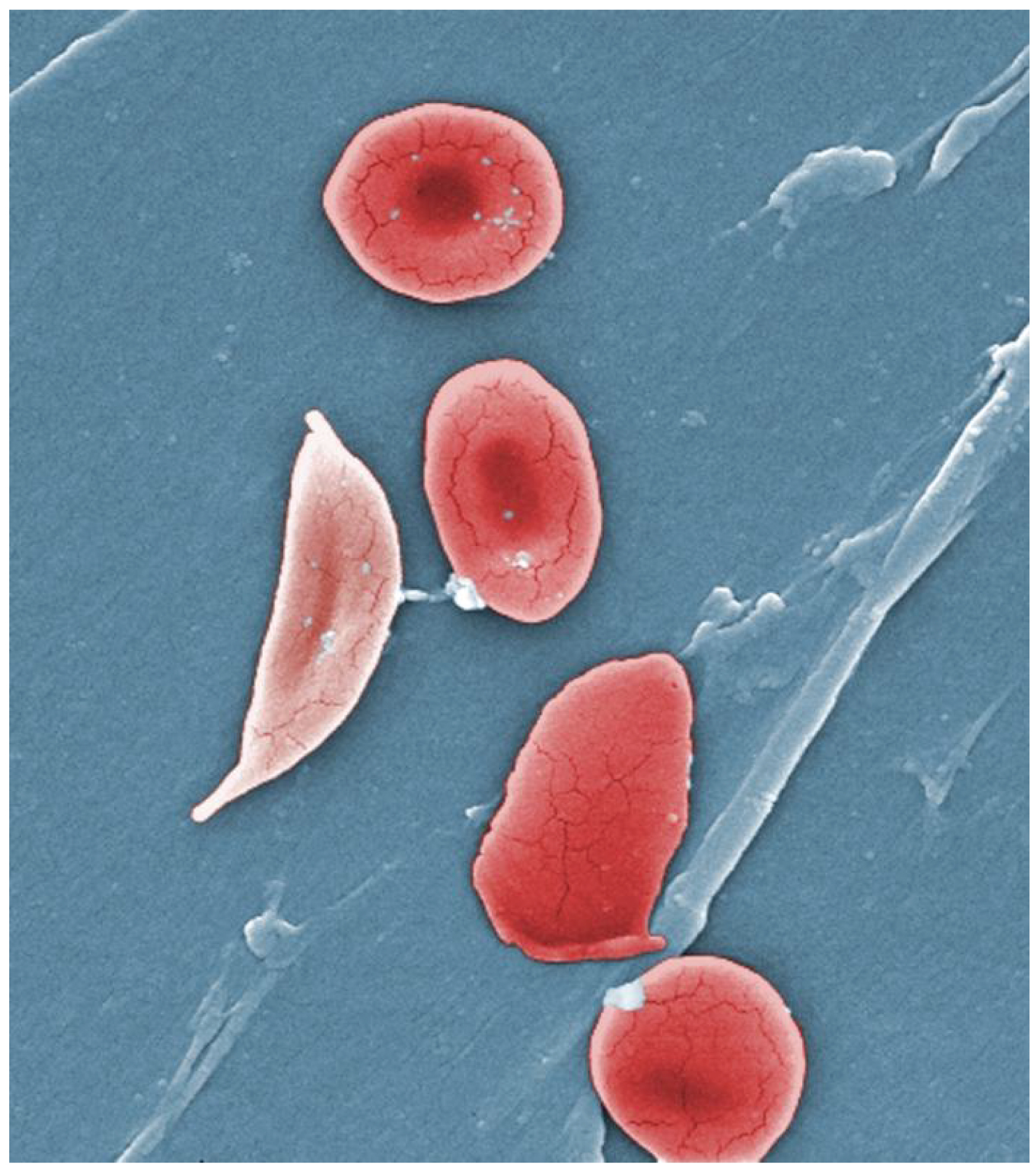
- A characteristic change in the shape of erythrocytes is seen in sickle cell disease (also referred to as sickle cell anemia). A genetic disorder, it is caused by production of an abnormal type of hemoglobin, called hemoglobin S, which delivers less oxygen to tissues and causes erythrocytes to assume a sickle (or crescent) shape, especially at low oxygen concentrations ((Figure)). These abnormally shaped cells can then become lodged in narrow capillaries because they are unable to fold in on themselves to squeeze through, blocking blood flow to tissues and causing a variety of serious problems from painful joints to delayed growth and fifty-fifty incomprehension and cerebrovascular accidents (strokes). Sickle jail cell anemia is a genetic condition especially found in individuals of African descent.
Sickle Cells
Sickle jail cell anemia is caused by a mutation in ane of the hemoglobin genes. Erythrocytes produce an aberrant blazon of hemoglobin, which causes the cell to take on a sickle or crescent shape. (credit: Janice Haney Carr)
- Iron deficiency anemia is the most common type and results when the amount of available iron is bereft to allow production of sufficient heme. This condition tin occur in individuals with a deficiency of iron in the diet and is especially mutual in teens and children as well as in vegans and vegetarians. Additionally, iron deficiency anemia may exist caused past either an disability to blot and transport iron or slow, chronic bleeding.
- Vitamin-scarce anemias by and large involve bereft vitamin B12 and folate.
- Megaloblastic anemia involves a deficiency of vitamin B12 and/or folate, and oft involves diets deficient in these essential nutrients. Lack of meat or a viable alternate source, and overcooking or eating insufficient amounts of vegetables may atomic number 82 to a lack of folate.
- Pernicious anemia is caused by poor absorption of vitamin B12 and is often seen in patients with Crohn's disease (a severe intestinal disorder oft treated past surgery), surgical removal of the intestines or stomach (common in some weight loss surgeries), intestinal parasites, and AIDS.
- Pregnancies, some medications, excessive alcohol consumption, and some diseases such as celiac disease are also associated with vitamin deficiencies. It is essential to provide sufficient folic acid during the early stages of pregnancy to reduce the hazard of neurological defects, including spina bifida, a failure of the neural tube to close.
- Assorted disease processes can also interfere with the production and formation of RBCs and hemoglobin. If myeloid stem cells are defective or replaced past cancer cells, there volition be insufficient quantities of RBCs produced.
- Aplastic anemia is the status in which there are scarce numbers of RBC stem cells. Aplastic anemia is frequently inherited, or it may be triggered by radiation, medication, chemotherapy, or infection.
- Thalassemia is an inherited condition typically occurring in individuals from the Center East, the Mediterranean, African, and Southeast Asia, in which maturation of the RBCs does not proceed normally. The most severe form is chosen Cooley's anemia.
- Lead exposure from industrial sources or even dust from paint chips of iron-containing paints or pottery that has not been properly glazed may too pb to destruction of the red marrow.
- Various illness processes too can pb to anemias. These include chronic kidney diseases often associated with a decreased production of EPO, hypothyroidism, some forms of cancer, lupus, and rheumatoid arthritis.
In contrast to anemia, an elevated RBC count is called polycythemia and is detected in a patient'south elevated hematocrit. It can occur transiently in a person who is dehydrated; when water intake is inadequate or water losses are excessive, the plasma book falls. As a result, the hematocrit rises. For reasons mentioned before, a mild form of polycythemia is chronic but normal in people living at high altitudes. Some aristocracy athletes train at high elevations specifically to induce this miracle. Finally, a type of bone marrow affliction called polycythemia vera (from the Greek vera = "true") causes an excessive production of immature erythrocytes. Polycythemia vera can dangerously elevate the viscosity of blood, raising claret pressure and making it more than difficult for the heart to pump blood throughout the body. It is a relatively rare affliction that occurs more ofttimes in men than women, and is more likely to be nowadays in elderly patients those over threescore years of age.
Chapter Review
The most abundant formed elements in blood, erythrocytes are red, biconcave disks packed with an oxygen-carrying chemical compound called hemoglobin. The hemoglobin molecule contains iv globin proteins bound to a pigment molecule called heme, which contains an ion of iron. In the bloodstream, iron picks upwards oxygen in the lungs and drops it off in the tissues; the amino acids in hemoglobin and then ship carbon dioxide from the tissues back to the lungs. Erythrocytes live only 120 days on average, and thus must exist continually replaced. Worn-out erythrocytes are phagocytized by macrophages and their hemoglobin is broken down. The breakup products are recycled or removed as wastes: Globin is broken down into amino acids for synthesis of new proteins; iron is stored in the liver or spleen or used by the bone marrow for production of new erythrocytes; and the remnants of heme are converted into bilirubin, or other waste products that are taken upward by the liver and excreted in the bile or removed by the kidneys. Anemia is a deficiency of RBCs or hemoglobin, whereas polycythemia is an backlog of RBCs.
Review Questions
Which of the following statements about mature, circulating erythrocytes is true?
- They have no nucleus.
- They are packed with mitochondria.
- They survive for an average of 4 days.
- All of the above
A molecule of hemoglobin ________.
- is shaped like a biconcave disk packed near entirely with iron
- contains four glycoprotein units studded with oxygen
- consists of four globin proteins, each jump to a molecule of heme
- can carry upwards to 120 molecules of oxygen
The production of salubrious erythrocytes depends upon the availability of ________.
- copper
- zinc
- vitamin B12
- copper, zinc, and vitamin B12
Aging and damaged erythrocytes are removed from the apportionment by ________.
- myeoblasts
- monocytes
- macrophages
- mast cells
A patient has been suffering for 2 months with a chronic, watery diarrhea. A claret exam is likely to reveal ________.
- a hematocrit beneath 30 percent
- hypoxemia
- anemia
- polycythemia
Critical Thinking Questions
A young woman has been experiencing unusually heavy menstrual bleeding for several years. She follows a strict vegan diet (no animal foods). She is at risk for what disorder, and why?
She is at take a chance for anemia, considering her unusually heavy menstrual bleeding results in excessive loss of erythrocytes each month. At the same time, her vegan diet means that she does non accept dietary sources of heme iron. The non-heme iron she consumes in plant foods is non equally well absorbed as heme atomic number 26.
A patient has thalassemia, a genetic disorder characterized by aberrant synthesis of globin proteins and excessive destruction of erythrocytes. This patient is jaundiced and is establish to have an excessive level of bilirubin in his claret. Explain the connectedness.
Bilirubin is a breakup product of the non-iron component of heme, which is broken from globin when erythrocytes are degraded. Excessive erythrocyte destruction would deposit excessive bilirubin in the blood. Bilirubin is a yellow pigment, and high blood levels can manifest as yellowed skin.
Glossary
- anemia
- deficiency of ruddy claret cells or hemoglobin
- bilirubin
- yellow bile pigment produced when iron is removed from heme and is farther broken down into waste products
- biliverdin
- greenish bile pigment produced when the not-iron portion of heme is degraded into a waste material production; converted to bilirubin in the liver
- carbaminohemoglobin
- compound of carbon dioxide and hemoglobin, and one of the ways in which carbon dioxide is carried in the blood
- deoxyhemoglobin
- molecule of hemoglobin without an oxygen molecule bound to it
- erythrocyte
- (as well, red claret prison cell) mature myeloid blood cell that is composed mostly of hemoglobin and functions primarily in the transportation of oxygen and carbon dioxide
- ferritin
- protein-containing storage course of atomic number 26 establish in the os marrow, liver, and spleen
- globin
- heme-containing globular poly peptide that is a constituent of hemoglobin
- heme
- carmine, iron-containing pigment to which oxygen binds in hemoglobin
- hemoglobin
- oxygen-carrying compound in erythrocytes
- hemosiderin
- poly peptide-containing storage form of fe found in the bone marrow, liver, and spleen
- hypoxemia
- below-normal level of oxygen saturation of claret (typically <95 per centum)
- macrophage
- phagocytic cell of the myeloid lineage; a matured monocyte
- oxyhemoglobin
- molecule of hemoglobin to which oxygen is bound
- polycythemia
- elevated level of hemoglobin, whether adaptive or pathological
- reticulocyte
- immature erythrocyte that may still comprise fragments of organelles
- sickle cell disease
- (also, sickle cell anemia) inherited blood disorder in which hemoglobin molecules are malformed, leading to the breakdown of RBCs that have on a characteristic sickle shape
- thalassemia
- inherited blood disorder in which maturation of RBCs does non go on normally, leading to abnormal formation of hemoglobin and the destruction of RBCs
- transferrin
- plasma protein that binds reversibly to atomic number 26 and distributes it throughout the trunk
Source: https://opentextbc.ca/anatomyandphysiologyopenstax/chapter/erythrocytes/
0 Response to "Can Polycythemia Vera Be Caused by Chemical Exposure"
Post a Comment